Spermatogenesis
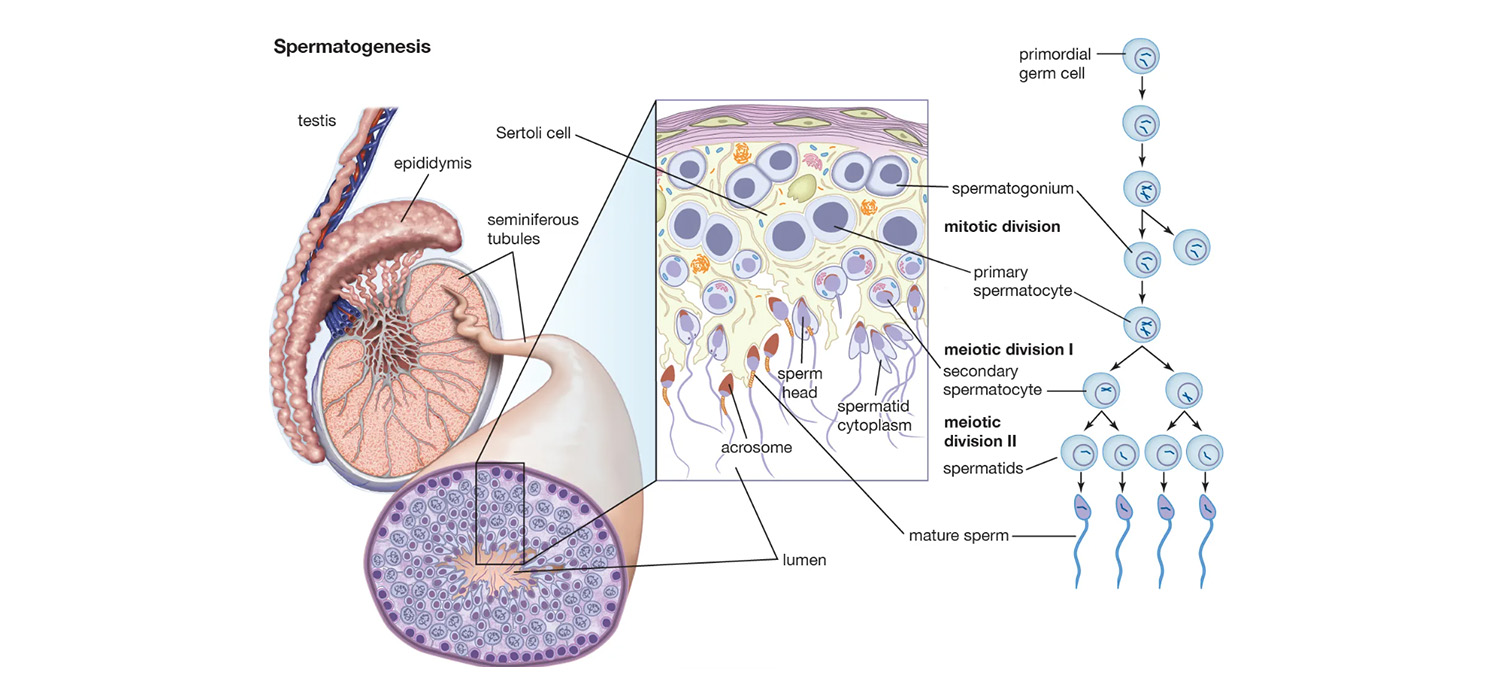
Spermatogenesis is the process by which haploid spermatozoa develop from the germ cells within the seminiferous tubules.
Embryology
Primordial germ cells originate in the extraembryonic mesoderm, posterior to the primitive streak, cross the dorsal mesentery and migrate to the gonadal ridge by about the 6th week of intrauterine life. The SRY gene in the Y chromosome activates TDF (Testicular Differentiating Factor) which activates the SOX 9 gene which helps in the formation of testes and development of the Sertoli cells.
The Sertoli cells-SC (Nurse cells/ sustentacular cells) are the epithelial cells in the gonadal ridge, and they are the first cells to differentiate in the testes, helping to co-ordinate testicular development. The SC surround the germ cells (GC) which are surrounded by peritubular myoid cells to form seminiferous cords that subsequently elongate to form seminiferous tubules. The Leydig cells are the other non-germ cells present in between the seminiferous tubules and are responsible for the production of testosterone.
Types of germ cells
The cross section of the seminiferous tubules reveals various categories of germ cells. These include spermatogonia, primary spermatocytes, secondary spermatocytes, spermatids and spermatozoa-from the basal to luminal part of the seminiferous tubule.
Spermatogonia are classified as type A and type B. Type A spermatogonia are further classified as Progenitor A dark (Ad)/ A0 spermatogonia which provide a continuous supply of stem cells for spermatogenesis. They are located near the basement membrane and can divide into both types of Ad and Ap cells. Progenitor and committed A pale (Ap)/A1 spermatogonia are similar in morphology but go on to become Type B spermatogonia and progress further. Type B spermatogonia divide further and produce the first cell of the second phase-the diploid preleptotene spermatocyte which crosses the tight junction between the SCs.The later maturation is into secondary spermatocytes, spermatids, and spermatozoa.
Mitosis and meiosis in the sperms
The primordial germ cells form gonocytes which are arrested in G0 phase till birth. After birth, the gonocytes develop into spermatogonia, which are inactive till 6-7 years and subsequently undergo mitosis. Further development occurs at puberty when the diploid primary spermatocytes that were arrested in meiosis I (preleptotene spermatocytes) resume meiosis and become haploid secondary spermatocytes. The live span of secondary spermatocytes is 1.1-1,7 days and is shortest.
The prophase lasts for about 3 weeks while the secondary oocytes.
Spermatogenesis
It takes about 74 days from the commitment of the Ap spermatogonium to the development of mature spermatozoa in humans.
Proliferation, meiosis, Spermiogenesis (differentiation) and spermiation are the four interdependent parts of spermatogenesis.
Ap spermatogonia become committed at circumscribed areas within each seminiferous tubules at an interval of 16 days and form clusters at various locations along the seminiferous tubules. Throughout the development the progeny of a spermatogonium stay joined together by cytoplasmic bridges till the late spermatid stage. Approximately 512 spermatids develop from one spermatogonium. 120-275 million spermatozoa are produced every day. There exists layers of generations of sperms with the earliest near the basement membrane. The multiple cohorts developing synchronously at a circumscribed area are called cellular associations.
Proliferation is mitosis for regeneration of progenitor Ad and Ap spermatogonia, formation of B spermatogonia and their further development into preleptotene spermatocytes.
Meiosis resumes with the primary spermatocyte (2n) which becomes haploid secondary spermatocyte, which undergoes meiosis II to become the haploid spermatid.
Spermiogenesis (differentiation) is the process by which the round spermatid changes to assume the shape of the human spermatozoa. There is cellular reorganization over 6 stages-Sa1, Sa2, Sb1, Sb2, Sc1 and Sc2. The three major modifications that occur are the condensation of chromatin to form the head of the sperm, the formation of the acrosomal cap and reorganization of the cytoplasm to form the tail and mid-piece.
Spermiation is the extrusion of the elongated spermatid into the lumen of the seminal tubules. FSH and testosterone act synergistically to aid spermiation.
Epididymal maturation
This refers to all the complex biochemical, biophysical, and ultramolecular changes that occur in the spermatozoa, bequeathing them with the ability to fertilize oocytes. Testicular fluid gets resorbed in proximal epididymis leading to concentration of sperms 10-100 times. Sperms negotiate the epididymis in 2-14 days and are stored in cauda, vas, seminal vesicles and ampullae before ejaculation. The changes that occur as sperms pass through the epididymis:
- Cytoplasmic droplets around the neck region are lost.
- Antigens needed to bind to the oocyte and for fertilization, which are secreted by testes, get activated as the sperms pass through the epididymis.
- Passage through epididymis provides motility to the sperms.
Blood testis barrier (BTB)
BTB is one of the tightest structures in the body. It consists of tight junctions between the adjacent Sertoli cells. It divides the seminiferous epithelium into the basal and the apical (ad luminal) compartments. Meiosis I and II, spermiogenesis, and spermiation all take place in a specialized microenvironment behind the BTB in the apical compartment, but spermatogonia renewal and differentiation and cell cycle progression up to the preleptotene spermatocyte stage take place outside of the BTB in the basal compartment of the epithelium. The functions of BTB include:
- Control passage of large molecules from the interstitial tissue and basal compartment to the lumen.
- BTB creates a specific composition of the fluid within the seminiferous tubules. The fluid is rich in androgens, estrogen, K+, inositol, glutamic and aspartic acid with traces of protein and glucose.
- BTB protects the spermatozoa from blood-borne toxins.
- BTB prevents antigenic products of spermatogenesis from entering into circulation.
Endocrine control of spermatogenesis
The hypothalamo-pituitary-testicular axis. Under the influence of GnRH from hypothalamus, the pituitary secretes FSH and LH. FSH acts on the Sertoli cells and results in secretion of Inhibin, MIS (Mullerian inhibiting substance) and ABP (androgen binding protein). LH acts on the interstitial cells of Leydig and causes secretion of testosterone. Testosterone and Inhibin, complete the circuit by their negative feedback at the hypothalamus and pituitary levels.
Genetic control
There are many genes involved in spermatogenesis and hence abnormalities in them can disrupt spermatogenesis. The AZF locus is located in the long arm of Y chromosome and microdeletions at AZF-a, AZF-b and AZF-c can cause impairment. Unbalanced translocations, pericentric inversions, Kallmann syndrome and Klinefelter syndrome are some pf the conditions with a genetic basis.
Effect of environment and lifestyle
Spermatogenesis occurs at a temperature of 32 degrees centigrade and hence raised temperature impacts spermatogenesis. Hot baths, insulated athletic supports, working in bakery/ foundry have all been implicated.
High BMI (>25) has been seen to reduce sperm count and motility by 25% and also cause reduction in testosterone.
Smoking and alcohol also affect sperm counts. Maternal smoking in pregnancy can impair spermatogenesis in male fetus.
Chronic use of medicines like sulphasalazine, cimetidine, antiepileptics, antibiotics can hamper spermatogenesis. Chemotherapy and radiotherapy can cause irreversible damage to spermatogenesis and hence sperm banking is recommended before definitive therapy.
Occupations requiring exposure to chemicals have been implicated in abnormal spermatogenesis. These include working with pesticides (DDT), glycol ethers (used as solvents in various procedures), heavy metals (lead, cadmium, mercury) and carbon-di-sulphide.
Understanding spermatogenesis helps in identifying the possible pathologies in conditions of abnormal sperm parameters and taking correct actions whenever feasible.
For more info, Visit : www.medlineacademics.com